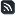What’s it all about, Flashfree? The Lowdown on Estrogen
I spend a lot of time and energy discussing the role of estrogen in menopausal symptoms. However, it occurred to me recently that readers might not truly understand the distinction between pre and post menopause and how estrogen levels interact with the body to cause a host of issues.
A few months ago, the Endocrine Society sent me an infographic that does a great job distilling this topic down to an understandable level. While I typically stay away from infographics, I like this for a number of reasons, including the fact that it is from a reputable source — the Hormone Health Network.
Read More
Give me a P!
P, as in “progesterone.”
I get a lot of questions on Flashfree about progesterone and whether or not it’s use is as dangerous as estrogen and combined hormone replacement. I have stayed away from the subject for some time now because the evidence has been pretty scarce, at least as far as natural progesterone goes.
Fortunately, a new review has appeared that provides a comfort level for a brief discussion. But is all progesterone alike? A good place to start, right?
Progesterone is a reproductive hormone produced after ovulation and during pregnancy; it is also produced by the nervous system and adrenal glands. It occurs naturally. Progestins are synthetic and manufactured outside the body and work differently depending on their structure. Progestins are often combined with estrogen therapy.
Although experts know that levels of progesterone decline as a woman transitions to menopause, the relation between this decrease and symptoms has not been well understood, thereby limiting practitioner’s — at least those who specialize in Western therapy — ability to make decisions regarding its use in ameliorating symptoms.
Following is what researchers learned by scouring the literature:
- In most cases, progesterone is prescribed as an oral pill ranging from less than 20 mg daily to more than 200 mg daily, or as a patch that delivers more than 50 mg daily. It is also available as a topical cream (the most reliable standardized formulation is manufactured by Emerita (Pro-Gest).
- High dose, oral progesterone and patch forms appear to provide the greatest benefits for lessening hot flashes.
- How well women respond to progesterone patches appear to be related to how severe their symptoms are; those women with the worst symptoms appear to gain the most benefit.
- Use of progesterone may help reduce bone loss, although more studies are needed.
- Use of topical progesterone may possibly help reduce the effects of aging on the skin by increasing elasticity.
- The oral forms have also been shown to hell women whose sleep is disrupted by increasing time spent asleep.
- Most importantly, low dose progesterone treatment, including oral agents, does not appear to increase heart disease risk. Even better? There is some indication that when combined with estrogen, progesterone may counteract negative effects int terms of breast cancer risk. Progesterone formulations also appear to be well tolerated, will only minimal side effects associated with their use.
The key take-away is the natural progestogens (progesterone) appear to safely alleviate many of the most troubling menopausal symptoms. The rub here is that the evidence for this be fit comes from small studies. Still, it is refreshing to learn that there are safe hormonal options for women who desire that route.
Read MoreHot flashes, Traditional Chinese Medicine & JQF – it is time for an Rx change?
I am a huge fan of Traditional Chinese Medicine (TCM), having used various preparations over the past 20 years for numerous ailments. These preparations have been prescribed by a practitioner who is licensed and degreed in TCM and my experience, albeit anecdotal, has been nothing but positive. In fact, I have been using a TCM formulation for several years now to help battle numerous symptoms and in combination with other prescribed herbs, I’ve mostly won. That’s one of the main reasons that I am always intrigued when I stumble upon well-designed studies that demonstrate benefit, like this one in the journal Menopause.
A bit of context
TCM and other similar philosophies emphasizes various body systems that together, form a network or grid connected by a meridien, if you will, as well as the relationship of the body to its social and natural environment. Its primary focus on maintaining health and enhancing the body’s ability to fight off disease. TCM will not focus, for example, on treating specific pathogens but rather, on addressing non-specific factors that create disturbances or imbalances within a certain network. TCM also examines how these imbalances may occur in unique parts of a specific system, such as the heart and blood vessels and small intestine (all of which are part of the heart system) and how they change over time. Western medicine, on the other hand, focuses primarily on treating morbidities, or symptoms related to various conditions and diseases. You can read morerabout TCM in this post from last September.
In TCM, menopausal symptoms are believed to be related to a decline in yin or yang in the kidneys. One of the oldest prescriptions in TCM to reinforce kidney energy is known as ‘Qing E Fang’ (QEF) and the first record of its use dates back to the year 960. The preparation is a combination of four key ingredients that are mediated through estrogen receptors and expression, hence, its potential usefulness in menopause. (These ingredients, in case you are interested, are Cortex Eucommiae (whose bark and leaf have an antioxidant effect) Fructus Psoraleae (commonly used for bone health and to treat estrogen associated diseases), Semen Juglandis and Rhizoma Garlic.)
About the research
For this study, researchers used a formulation based on QEF known as Jiawei Qing’s Fang or, JQF, which used two of the four herbs (Cortex Eucommiae, Fructus Psoraleae) mentioned above. In JQF, they are combined with the herb, Salviae Miltiorrhizae, that has been used specifically to treat gynecological disorders and has properties that are similar to SERMs, drugs that mimic estrogen but are theoretically safer. The 72 women participating in the study were all perimenopausal and had severe and frequent hot flashes; they were randomly instructed to take JQF or placebo daily over 8 weeks. For a month thereafter, symptoms were evaluated using a menopause measure that looks at vasomotor, physical, psychosocial and sexual health). The women also kept daily diaries to record the frequency and severity of their flashes.
Notably, this is one of the first times that perimenopausal women have been the focus of a study that involves TCM. Moreover, in this study, researchers selected a formulation that worked on the kidney and also contained an herb that not only mimics SERMS but also improves blood circulation. And the findings? Women who randomly took JQF not only experienced significant improvements in their hot flashes, but also reported improvements in quality of life in terms of the impact of vasomotor and physical symptoms. Moreover, because Salviae Miltiorrhizae works on blood circulation, they also had reductions in the blood fats (triglyerides) which means that the formulation might also be beneficial in terms of heart disease prevention in menopausal women.
Is it time for a change of Rx for the change? Should you be seeking out a practitioner who can prescribe JQF and advise you on its best use for you? Or is it too early? JQF was well tolerated, although two women were found to have liver measures negatively affected in ways that did not require any medical intervention but did raise a few red flags and calls for more study. JQF represents a seemingly scientifically proven alternatives to hormone therapy. It’s refreshing to watch TCM come out of Asia and be increasingly incorporated into Western philosophy.
Estrogen and urinary incontinence: is there a link?
One of the most common and (and yet unspoken about) conditions in women is urinary incontinence (UI) or problems with bladder control. Defined as the involuntary loss of urine – either due to a weakening of the pelvic floor muscles and in association with pressure on the bladder (stress urinary incontinence) or due to unknown causes and associated with an uncontrollable urge to pass urine, frequency and nighttime awakening (urge urinary incontinence or overactive bladder) – urinary incontinence is most definitely associated with aging. In fact, roughly 15 million women in the U.S. have stress urinary incontinence and about 20 million, overactive bladder.
There are a multitudes of risk factors for urinary incontinence and they range from weight, vaginal deliveries and pelvic surgery to alcohol use and of course, as mentioned, growing older. However, why is menopausal status also a risk factor?
One word: estrogen.
Indeed, results from the infamous Women’s Health Initiative study demonstrated that women who were randomized to combination hormone therapy or estrogen only were at increased risk for worsening urinary incontinence symptoms or for developing urinary incontinence after only one year of use. However, like other data from this study, questions have been raised with regard to the findings, namely that they are not applicable to the general population. And yet, it is critical to learn if using hormone therapy increases urinary incontinence risk; these conditions significantly affect quality of life and at their severest, limit physical and social activities, limit intimacy and other relationships, limit work productivity and affect overall wellbeing.
Rather than generalize, however, it’s important to take a close look at ethnically diverse populations of women in the community and tease out if there are any specific factors related to estrogen use that increase incontinence risk. That is exactly what a group of researchers did recently, when they examined a group of 167 women in menopause who had been surveyed in 1993, found to have no urinary incontinence and then reinterviewed eleven years later in 2004. In this study, which was published in Menopause journal,the researchers specifically evaluated if the women had used estrogen and if so, for how long (i.e. less than five years or more than five years). The findings? Although none of the women reported having urinary incontinence issues at the first interview, just over a decade later, 28% reported that they had developed urinary incontinence and almost 19%, that they developed urinary incontinence that resulted impacted their ability to function (e.g. avoiding social gatherings, not visiting friends or going to church, or avoiding traveling, shopping or physical activities). What’s more, of the women surveyed who reported that they had used estrogen for more than five years, 15% developed new cases of urinary incontinence with an associated loss of function.
According a related piece in Reuters, the study’s lead investigator says that they didn’t take into account how much estrogen the women were using or if they used it in conjunction with progesterone, so there are weaknesses in the study. Still, it does appear that taking estrogen for more than five years may significantly increase the risk for bladder control issues. The next piece of the puzzle is discovering why it affects bladder function in the first place.
Bladder control issues are serious business. Yet another reason to speak to your doctor before moving forward on hormone therapy. Your move – worth the risk?
Read MoreMenopause: the symptom? Or, the disease?
A few years ago, I ran across the following story on the BBC:
“Woman’s Death Blamed on Menopause.”
“A woman who refused to take hormone replacement therapy died while suffering a menopausal episode, an inquest had heard. Margaret Drew…was killed when she walked out of her family home on to a nearby railway line and was hit by a train…There is no trigger to this at all, except hormones making her do things that she normally wouldn’t do, Dr. Carlyon [Cornwall Coroner) concluded…”
Menopause. The silent killer. Oh really? Drew’s husband claims that his wife was “delightful, lovely and friendly” 99% of the time; the other 1% she’d become “totally irrational.” Yet, she refused to try HRT, he says. On the day of her suicide, he said that his wife was “clearly angry about something.”
Something.
Obviously, the conclusion is that that the “something” is hormones. This reminds me of vintage advertising copy that conveys the simple message that a pill a day can cure all that ails, wipe away the tears, mood swings and instability so that women can “transition without tears” (or better yet, without killing themselves).
Notably, a search in the National Library of Medicine’s PubMed database turned up only one recent study specifically dealing with suicide ideation across reproductive stages. In it, researchers compared data in 8,794 women, and found an increased risk of thinking about suicide among women during perimenopause, not before or after entering menopause. These findings remained after controlling for risk factors such as anxiety and mood disorders. HOWEVER, the researchers noted that the study design did not allow them to form any definitive conclusions about the specific reasons for thinking about suicide.
Another search yielded information that the risk for a major depression increases during perimenopause, primarily as the direct result of vasomotor symptoms. The same does not hold true for women before menopause begins or once they enter menopause. Note that while major depression is a risk factor for suicide, not everyone who is depressed will actually kill themselves.
So, are hormonal fluctuations the sole cause of such deep unhappiness that women want to kill themselves?
Interestingly, just a week after the menopause/train suicide story hit the interwebz, a rather controversial set of data also emerged: since 1972, women’s overall level of happiness has dropped. These findings held true regardless of child status, marital status and age. Researcher Marcus Buckingham, writing in the Huffington Post, said that women are not more unhappy than men because of gender stereotyping and related attitudes, due to working longer hours or because of the inequality of housework/responsibilities at home, but rather, the hormonal fluctuations of menopause may be to blame. What’s more, he leaves us hanging so we’ll tune in for part two of the piece to learn the true cause of our declining happiness or better yet, read his book (which evidently guides women through the process of finding the true role that they were meant to play in life).
Importantly, reactions to this study (and various pundits’ assessment of it) have been mixed. One of the most poignant comments I’ve read asks the question “how is happiness measured? What does it mean?”
I have no idea what caused Mrs. Drew to walk into a train and kill herself. Perhaps she was depressed. Clearly she was suicidal.
I have no idea why research shows that women are less happier than they were three decades ago.
However, is menopause the cause? Aren’t these conclusions an example of how the Menopause Industrial Complex perpetuates societal myths that menopause is a disease that requires treatment? That as women, our attitudes, belief systems and actions are hormonally-based and driven? That we are hysterical beings who need guidance on how to find our way and fulfill our dreams, realize our paths, but only if we calm down?
Feeling angry? Blame it on menopause. Unhappy? Blame it on menopause. Not realizing your dreams? Blame it on menopause. Overworked, overstressed, undervalued? Blame it on menopause.
Blame it on menopause.
I don’t know about you but I’m tired, tired of hearing that menopause is not the symptom but the disease. Isn’t it time we start fighting back?
Read More